Background – Endometrial cancer and this trial
Uterine-serous-carcinoma, USC, is an aggressive variant of endometrial cancer.
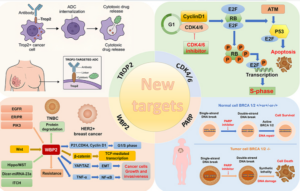
This phase II trial evaluated Carboplatin+Taxol versus Herceptin +Carboplatin+Taxol in patients with advanced or recurrent uterine serous carcinomas that express HER2, HER2+. Eligible patients had stage III to IV metastatic or recurrent disease.
The addition of Herceptin improved both the time until worsening of the disease and survival, with the greatest benefit observed for patients with advanced or metastatic disease.
Results
- 61 patients were enrolled to the study
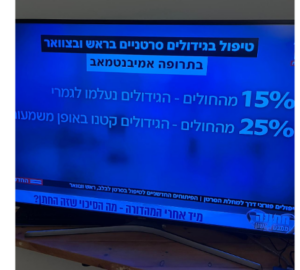
- Median time until worsening of disease was 8.0 months in the Carboplatin+Taxol group versus 12.9 months in the Herceptin+Carboplatin+Taxol group
- Median time until worsening of disease was 9.3 months versus 17.7 months in the Herceptin group among 41 patients with stage III to IV disease for whom this was their primary treatment
- Median time until worsening of disease was 7.0 months versus 9.2 months in the Herceptin group among 17 patients with recurrent disease
- Median survival was higher in the Herceptin group – 29.6 months versus 24.4 months.
- Side effects were not different between groups
Conclusions in Endometrial cancer
Addition of Herceptin to Carboplatin+Taxol increased the time until the disease worsened and longevity in women with advanced/recurrent HER2+ USC, with the greatest benefit seen for the treatment of stage III to IV metastatic disease.
The best treatment for a cancer patient is to get the most advanced cancer drugs in advanced stages of development. There, the hope and the chance to extend life go far beyond the standard protocols.
Contact us to find out what is the best treatment for YOU
TRIAL•IN Pharma
Because we, do not give up on life!
Contact us 24/7 –
Call center +44.2082.426.039
A clinical trial is a study where human beings receive the treatment, and there are approx.
Under the definition of “NEW THERAPY”, are: a new drug, a new indication for a known drug, or a new technique of performing a surgery, or providing radiation therapy. A new therapy may be a treatment using a new technique (such as gene manipulation) or a new medical device.
20,000 cancer clinical trials are available to cancer patients worldwide. Less than a few percent of these studies are open in each country.