Home » Glioblastoma
Glioblastoma
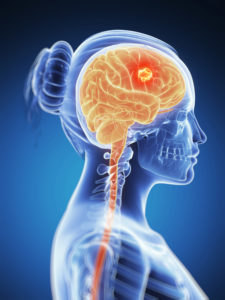 About Glioblastoma
About Glioblastoma
Glioblastoma Multiforme, or GBM, is a type of cancer which originates in the brain and made of brain cells known as “gliomas.”
Gliomas are neuron supporting cells, constituting a part of the nervous system.
Glioblastoma tends to develop into a star shaped formation. They are particularly aggressive tumors with the potential to grow fast and spread to other parts of the brain relatively quickly.
Glioblastoma creates its own independent blood supply which feeds it, promoting its growth and even enabling it to invade additional areas of the brain and establish more foci, hence the name “multiforme.”
GBM is a stage 4 cancer and constitutes about 50% of all brain tumors among patients aged 18 and older. Glioblastoma does not metastasize outside the brain.
The existing therapies used for glioblastoma are not curative, and this is a source of the need for innovative and effective treatment strategies in order to fight the disease.
The National Cancer Institute, NCI, highlights the fact that for a certain group of glioblastoma patients, the best treatment option is to join one of the many clinical trials existing worldwide aiming to increase their chances of therapeutic success.
Glioblastoma Multiforme, GBM, Symptoms
With its speedy growth rate, and the fact that the skull is incapable of expanding, internal pressure builds up inside the brain.
The pressure causes an array of symptoms based on the location of the tumor.
Symptoms may include ongoing and permanent headache, seizures, vomiting, confusion or cognitive complications, mood swings or behavioral changes, double or blurred vision, speech difficulties, and more.
Recurrent Glioblastoma
In most cases, despite these treatments, Glioblastoma tends to recur and re-grow. This stage is known as rGBM, recurrent Glioblastoma.
At this point the options for conventional treatments are fewer.
When this occurs, treatments the patient has already received are often repeated since no other options exist among standard protocols.
Treatment Goals
The treatment objectives in Glioblastoma are to slow down and control the tumor’s growth rate and assist the patient in maintaining a good quality of life as much as possible.
Standard Treatments
A number of therapies are possible, and many patients shall receive more than one type of therapy.
The primary challenge in developing an effective drug for GBM is the “BBB,” the Blood Brain Barrier.
This is the membrane which envelopes the brain and prevents penetration or entry of various chemicals into the brain, and that also includes drugs.
Five main treatments exist for Glioblastoma, and many patients receive more than one type of treatment.
Surgery
Surgical procedures aim to remove as much tumor mass as possible from the brain without damaging the brain’s normal functioning.
Success is marked by the removal of the entire tumor but this is not always possible, since the tumor is often multibranched, shaped like a star, and cannot be fully removed.
If the tumor is located in a highly risky area of the brain, the patient is considered inoperable in order to avoid problematic or irrevocable brain damage.
Usually after the surgical procedure and recovery, patients then continue onto radiation therapy and chemotherapy.
Radiotherapy
This involves use of radiation at higher levels of energy than X-rays, gamma rays, neutrons, protons and other sources.
Radiation therapy aims to kill the cancer cells remaining in the post-operative brain.
Radiation may also cause decelerated growth of the localized tumor remaining in the brain, or local recurrence.
Chemotherapy
Chemotherapy is a medicated therapy which aims to shrink and erase cancerous cells and allow control over the disease.
Chemotherapy carries serious side effects, while its effectiveness is insufficient, and stands at around 20% for GBM.
Chemotherapy’s impact has a range of side effects such as hair loss, loss of appetite, changes to bowel movements, fatigue, infection, hemorrhaging, incontinence, pain, and more.
Common chemotherapy treatments for GBM include:
- Temodar®, Temodal® (Temozolomide) – The is the most common form of chemotherapy prescribed by physicians for most GBM patients. It constitutes the standard treatment, is administered as pills, and is known for many years as the Glioblastoma treatment.
- BCNU – Long standing form of chemotherapy
- Gliadel®, (Carmustine) – a type of implant infused with BCNU. It is placed on the area in the brain during operation. Since the implant is biodegradable, there is no need to remove it in a repeat surgical procedure.
Biological Treatments
- Avastin®(Bevacizumab) – Biological antibody which detrimentally affects the tumor’s ability to promote blood vessels growing into the tumor from its environment. Without blood vessels feeding the tumor, it cannot grow and proliferate. Basically, the tumor is “starved” to death. Among GBM patients the rate of response to Avastin stands around 20%, the length of response time for those who respond to treatment is limited however, life expectancy is almost tripled.
Electrical fields
TTF (Tumor Treating Fields), Optune®
Both treatments involve positioning electrodes directly on the patient’s skull.
Electrical fields are directed to the brain tumor. In this way, the electric fields do not damage healthy cells but definitely disrupt the process of cancerous cells dividing and multiplying, and eventually brings about the eradication of cancerous cells.
This treatment has been proven to hold a clear advantage when patients are simultaneously treated with electrodes and chemotherapy.
The Optune® device constitutes a breakthrough option in this field, developed by an Israeli company Novocure®.
These are the standard treatments which may lead to some degree of tumor regression and relief of symptoms for a limited period among a small number of GBM patients.
 Advanced Cancer Drugs, Breakthrough Treatments and Clinical Trials from Around the World for Glioblastoma, GBM, Patients
Advanced Cancer Drugs, Breakthrough Treatments and Clinical Trials from Around the World for Glioblastoma, GBM, Patients
The Facts
- The existing therapies used on glioblastoma are not curative, and this is a source to the need for innovative and effective treatment strategies in order to fight the disease.
- The National Cancer Institute, NCI, highlights the fact that for a certain group of glioblastoma patients, the best treatment option is to join one of the many clinical trials existing worldwide aiming to increase their chances of therapeutic success.
- Currently, every patient will be offered the standard protocols described above. Sometimes the oncologist may suggest integrating these therapies with trials carried out in the institute.
The Opportunity
- Advances in cancer therapies may be found in clinical trials prescribing cutting edge, innovative drugs, some of which have already been recognized by the US Food & Drug Administration, the FDA, as “breakthrough drugs” and require further information to be approved.
- An entire world of clinical trials, Compassionate drugs and advanced new cancer treatments are available worldwide.
- It is important to know and have access to cutting edge cancer treatments that best match the unique medical condition of the patient and increase chances in winning the fight against compared to the standard care offered.
Example demonstrating the contribution of clinical trial to the lives of GBM patients
In October 2017, the initial results of a clinical breakthrough trial were published in the USA. They involved genetic treatment of Glioblastoma, and proved that 25% of patients receiving this treatment lived more than 3 years from the start of it!
Several fascinating points arise from this research:
- The achieved advantage was not specific mutation or biomarker dependent in the tumor’s cells. This means that GBM patients in general can benefit from this treatment, rather than only those patients who have specific mutation-based tumors
- The study found that the therapy not only kills cancer cells in the brain but also enhances the patient’s own immune system and reinforces the immune system’s ability to identify and attack cancer cells. This process increases the treatment’s chances of success
- It was also found that the treatment is Glioblastoma focused, a clear advantage vis-à-vis typical adverse effects of chemotherapy resulting from the whole body being affected by the standard chemotherapy treatment
- This study has completed its recruitment of new patients but another similar study is currently underway with the same treatment. Reports so far indicate that out of 23 rGBM patients participating in the study, full regression was observed in 5 patients. Regression lasted for three years on average. In 5 other patients, disease progression was halted.
Note to remember!
Not every patient is eligible to enter a specific clinical trial. The eligibility conditions must be fully met and each case is reviewed separately to ensure that the clinical trial matches to the specific cancer patient and vice versa.
References:
https://www.fda.gov
https://www.ema.europa.eu/
https://www.health.gov.il/Pages/HomePage.aspx
https://www.aans.org/Patients/Neurosurgical-Conditions-and-Treatments/Glioblastoma-Multiforme
https://www.abta.org/wp-content/uploads/2018/03/about-brain-tumors-a-primer-1.pdf
https://www.webmd.com/cancer/brain-cancer/what-is-glioblastoma#1
https://www.cancerresearchuk.org/
novocure.com
Dear Reader
This information is provided as a source of knowledge and does not constitute a medical consultation. For professional medical advice, see your attending physician. Information regarding drugs and commercial names belongs to the pharmaceutical companies.
To check if our service suits your case
We need to talk

Improve therapeutic outcomes, prolong life and quality of life, are our main business.
We support access to and expand cancer treatments beyond the standard of care, with the most advanced and innovative treatment options in the world, personally matching the individual cancer patient’s medical condition and with the support of top tier oncologists.
Article categories
קטגוריות המאמרים
Popular topics