Home » DIPG (brain stem cancer)
DIPG (brain stem cancer)
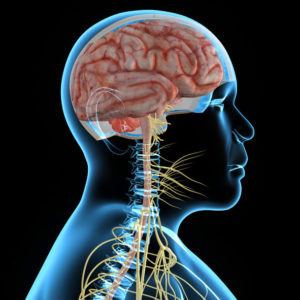
DIPG – The disease develops in the brain stem that is located at the base of the brain and connects the brain to the spine.
About DIPG , Brain Stem Cancer
DIPG (brain stem cancer), Diffuse Intrinsic Pontine Glioma, is a type of cancer which originates in the brain and made of brain cells known as “gliomas.”
Gliomas are neuron supporting cells, constituting a part of the nervous system.
The disease develops in the brain stem that is located at the base of the brain and connects the brain to the spine.
The brain stem is responsible for basic activities: vision, hearing, speech, walking, eating, breathing, pulse and more.
DIPG (brain stem cancer) is a pediatric disease. The average age of diagnosis is 4 to 11, and the disease accounts for about 10-15% of all brain tumors among children. Some adults have the disease as well.
Brain stem tumors are not autopsied and the disease is considered aggressive by affecting the ability to eat, drink, and move. The tumor grows, develops and presses the nervous system, disrupting basic functioning. In advanced stages, the tumor interferes with breathing and pulse, leading to death.
The existing therapies used for DIPG are not curative, and this is a source of the need for innovative and effective treatment strategies in order to fight the disease.
The National Cancer Institute, NCI, highlights the fact that for a certain group of DIPG patients, the best treatment option is to join one of the many clinical trials existing worldwide aiming to increase their chances of therapeutic success.
DIPG Symptoms
In the early stages of the disease the symptoms are mainly in the face:
- Problems with vision and movement of the eyes, double vision, drooping eyelids.
- Hearing problems.
- Problems eating, difficulty eating and chewing.
- Weakness of muscles and organs e.g. drop of face, difficulty lifting hands or legs, lack of balance.
The advanced stages of the disease will be
- Headaches and edema.
- Nausea and vomiting.
DIPG Diagnosis
The diagnosis is usually done using CT or MRI, and not by taking a biopsy, which is the precise way to ensure that the lesion is cancerous.
These tumors are difficult to take biopsy from by surgery because of their location such a procedure may pose a risk to the patient.
Treatment Goals
The treatment objectives are to slow down and control the tumor’s growth rate and assist the patient in maintaining a good quality of life as much as possible.
Standard Treatments
There are almost no treatments for the disease.
The primary challenge in developing an effective drug for DIPG is the “BBB,” the Blood Brain Barrier.
This is the membrane which envelopes the brain and prevents penetration or entry of various chemicals into the brain, and that also includes drugs.
Surgery
Due to the problematic location of the tumor, it is usually impossible to operate. This is a dangerous area of the brain and surgery can cause a dangerous and irreversible injury.
If so, the purpose of the surgery is to remove as much tumor mass from the brain as possible without damaging the brain’s normal functioning.
Success is marked by the removal of the entire tumor but this is not always possible, since the tumor is often multibranched, shaped like a star, and cannot be fully removed.
If the tumor is located in a highly risky area of the brain, the patient is considered inoperable in order to avoid problematic or irrevocable brain damage.
Usually after the surgical procedure and recovery, patients then continue onto radiation therapy and chemotherapy.
Radiotherapy
This involves use of radiation at higher levels of energy than X-rays, gamma rays, neutrons, protons and other sources.
Radiation therapy aims to kill the cancer cells remaining in the post-operative brain.
Radiation may also cause decelerated growth of the localized tumor remaining in the brain, or local recurrence.
Chemotherapy
Chemotherapy is a medicated therapy which aims to shrink and erase cancerous cells and allow control over the disease.
Chemotherapy’s impact has a range of side effects such as hair loss, loss of appetite, changes to bowel movements, fatigue, infection, hemorrhaging, incontinence, pain, and more.
Advanced Cancer Drugs, Breakthrough Treatments and Clinical Trials from Around the World for DIPG Patients
Facts
- The existing therapies used on DIPG are not curative, and this is a source to the need for innovative and effective treatment strategies in order to fight the disease.
- The National Cancer Institute, NCI, highlights the fact that for a certain group of DIPG patients, the best treatment option is to join one of the many clinical trials existing worldwide aiming to increase their chances of therapeutic success.
- Currently, every patient will be offered the standard protocols described above. Sometimes the oncologist may suggest integrating these therapies with trials carried out in the institute.
The Opportunity
- Advances in cancer therapies may be found in clinical trials prescribing cutting edge, innovative drugs, some of which have already been recognized by the US Food & Drug Administration, the FDA, as “breakthrough drugs” and require further information to be approved.
- An entire world of clinical trials, Compassionate drugs and advanced new cancer treatments are available worldwide.
- It is important to know and have access to cutting edge cancer treatments that best match the unique medical condition of the patient and increase chances in winning the fight against compared to the standard care offered.
What Does the Future Hold for DIPG?
US researchers found that nearly 80% of DIPG have mutations in the gene responsible for the formation of a protein called “histone H3”. In animal trials, it was observed that drugs inhibiting PRC2 and BET helped stop the activity of histone H3, and extended life.
A second study examining the role of the enzyme PRC2 found that a drug called “tazmetostat” taken orally and inhibits PRC2 reduced the growth of DIPG cells.
Further research is needed on these treatments to test their actual activity on humans, but researchers are excited about their potential to prolong life and improve the quality of life of these young cancer patients.
Note to remember!
Not every patient is eligible to enter a specific clinical trial. The eligibility conditions must be fully met and each case is reviewed separately to ensure that the clinical trial matches to the specific cancer patient and vice versa.
References:
https://www.fda.gov
https://www.ema.europa.eu/
https://www.health.gov.il/
https://www.aans.org/
https://www.abta.org/
https://www.webmd.com/
Dear Reader
This information is provided as a source of knowledge and does not constitute a medical consultation. For professional medical advice, see your attending physician. Information regarding drugs and commercial names belongs to the pharmaceutical companies.
To check if our service suits your case
We need to talk

Improve therapeutic outcomes, prolong life and quality of life, are our main business.
We support access to and expand cancer treatments beyond the standard of care, with the most advanced and innovative treatment options in the world, personally matching the individual cancer patient’s medical condition and with the support of top tier oncologists.
Article categories
קטגוריות המאמרים
Popular topics