Home » Case Studies or Case Reports
Case Studies or Case Reports
What is a Case Report?
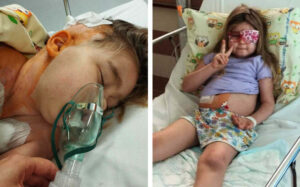
In medicine, a case report is an article detailing symptoms, signs, diagnosis, treatment and follow-up on a single patient only. Case reports have great value and significant one in cancer care. They shed light in the diagnosis and treatment of a disease.
Case reports may present the demographic features of the patient, but usually, they describe an unusual or intriguing case observed on a single patient, for example, a profound response of a cancer patient to a drug used to treat a different disease and there are no clinical trials with the same drug in cancer or for example, an unprecedented response to a certain drug when given to a cancer patient with a rare type of cancer where there is no standard protocol for this specific type of cancer.
In order to present a case report in a scientific congress or have it published in the professional literature, it has to meet strict criteria, for example:
- The case should bring a new treatment option and contain a medical description never seen before
- The case present no unusual side effects or severe interactions between drugs
- The disease did not worsen during the treatment, did not progressed
- No new and different disease has occurred
What do we do with case reports?
At TRIAL•IN Pharma publications of case reports are mapped and we identify those matching the patient based on the current medical background and his or her clinical history. We equip the patient with personally matched information, in a clear and concise manner with which he or she continues to a consultation with the treating oncologist to decide together on the best treatment.
TRIAL•IN Pharma holds a unique specialty which combines identification and analysis of case reports and supports stage 4 metastatic cancer patients access to advanced treatments and clinical trials around the world.
To check if our service suits your case
We need to talk

Improve therapeutic outcomes, prolong life and quality of life, are our main business.
We support access to and expand cancer treatments beyond the standard of care, with the most advanced and innovative treatment options in the world, personally matching the individual cancer patient’s medical condition and with the support of top tier oncologists.
Article categories
קטגוריות המאמרים
Popular topics