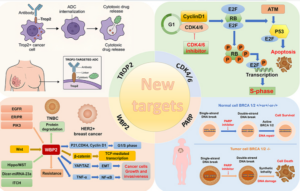
About Non Small Cell Lung Cancer, NSCLC
Lung cancer is the leading cause of cancer death globally. Each year 1.76 million people die as a result of the disease; this translates into more than 4,800 deaths worldwide every day. Lung cancer can be broadly divided into two major types: NSCLC and small cell lung cancer. NSCLC is the most prevalent type, accounting for around 85% of all lung cancers. NSCLC comprises non-squamous and squamous-cell lung cancer.
Despite recent advances in the treatment of NSCLC, there is still a need for new options to support a tailored approach to treatment of this complex disease.
About the EMA and the positive opinion
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion for Tecentriq®, atezolizumab, in combination with standard chemotherapy used to treat lung cacner – carboplatin and Abraxane®, for the first-line treatment of adults with metastatic non-squamous non-small cell lung cancer, NSCLC, who do not have EGFR mutant or ALK-positive NSCLC. A final decision regarding the approval of this Tecentriq-based combination is expected from the European Commission in the near future.
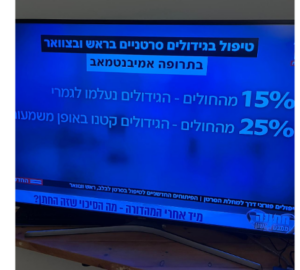
This decision is based on results from the Phase III IMpower130 study, which demonstrated that the Tecentriq combination therapy helped people live significantly longer, 18.6 months, compared with chemotherapy alone, 13.9 months; The combination also significantly reduced the risk of disease worsening or death, 7.0 months, compared with chemotherapy alone, 5.5 months;
Tecentriq in combination with chemotherapy shrank tumors in 49.2% of people compared with 31.9% of people on chemotherapy alone.
The median duration of response for people receiving Tecentriq in combination with chemotherapy was 8.4 months compared with 6.1 months for people on chemotherapy alone.
The safety profile of the Tecentriq combination therapy was consistent with that observed in previous studies.
Serious adverse events were reported in 73.2% of people receiving Tecentriq plus chemotherapy compared to 60.3% of people receiving chemotherapy alone.
In January 2019, the FDA accepted a supplemental biologics license application for atezolizumab, carboplatin, and Abraxane as a first-line treatment for patients with metastatic nonsquamous NSCLC. The FDA is scheduled to make a decision by September 2, 2019.
About the IMpower130 study
IMpower130 is a Phase III, multicentre, open-label, randomized study evaluating the efficacy and safety of Tecentriq in combination with carboplatin and Abraxane versus carboplatin and Abraxane alone for chemotherapy-naïve patients with stage IV non-squamous NSCLC. The study enrolled 723 people.
People who received carboplatin and Abraxane alone were given the option to crossover to receive Tecentriq as monotherapy until disease progression.
There is no doubt that patients who participated in the study are the real winners – This treatment worked, was successful and they received it long before its official approval and reimbursement by the world health authorities.
About Tecentriq (atezolizumab)
Tecentriq is an immunotherapy drug, a monoclonal antibody designed to bind with a protein called PD-L1, which is expressed on tumor cells and immune cells.
The best management for any patient with cancer is receiving the most innovative cancer drugs in advanced stages of development. There, the hope and chances to live longer go far beyond the standard protocols.
TRIAL•IN Pharma
Do not give up on life!
Contact us 24/7 for free service matching:
Call center +44.2082.426.039