Background – New option for Liver cancer
Hepatic Arterial Infusion Chemotherapy, HAIC, is a medical procedure that delivers chemotherapy directly to the liver and widely used.
Toripalimab an antibody against PD-1. Toripalimab is approved in China for other indications.
Lenvima is a cancer medicine that interferes with the growth and spread of cancer cells in the body. Lenvima is approved for different indications among is as the first treatment for advanced liver cancer.
Study showed that adding Toripalimab and HAIC to Lenvima represents a potential new treatment option for patients with advanced liver cancer in the first line setting, as it yielded robust, durable responses.
About the Study, LTHAIC study
This single-arm, phase II study included treatment-naive patients with advanced HCC (NCT04044313).
Patients initiated Lenvima 3-7 days prior to initial HAIC and then continued to receive Lenvima but with Toripalimab and HAIC with chemotherapy which was the FOLFOX regimen.
The trial enrolled 36 patients.
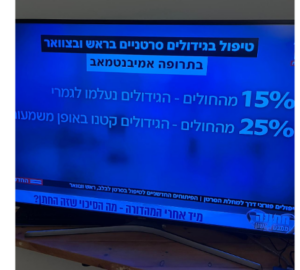
Study Results
- At 6 months from the start of the treatment, the disease did not progress in 80.6% of patients
- The median time the disease had progressed was 10.5 months
- About two-third of patients had their disease shrunked or stopped from growing
- More impressive was the fact that at the time of the analysis, the median survival was not reached
- In about 14% of patient, not disease was seen in the scans!
- Patients experienced a median duration of response of 12.1 months
Side Effects
Serious side effects occurred in 72.2% of patients, the most common being thrombocytopenia (13.9%), elevated aspartate aminotransferase (AST; 13.9%) and hypertension (11.1%).
Moreover, the most common any side effects were hypoalbuminemia (69.4%), elevated AST (66.7%), and elevated alanine aminotransferase (ALT; 58.3%).
However, all side effects were expected and managed, with no treatment-related deaths occurring on the study.
Conclusion
Adding Toripalimab and hepatic arterial infusion chemotherapy to Lenvima yielded robust, durable responses for patients with advanced liver cancer.
Talk to us so see if we can help you to actually get the most advanced treatments
TRIAL•IN Pharma
Because we, do not give up on life!
Contact us 24/7 –
Call center +44.2082.426.039
More information about liver cancer>>
About Liver Cancer
Liver cancer, also called “Hepatocellular carcinoma”, HCC, has several types based on the type of cells which turn cancerous.
Liver cancer is aggressive, since most of the patients are diagnosed in an advanced stage, where the disease is considered incurable, for the most part.
The patient’s life expectancy is poor and currently stands on 3-5 year; this is with the support of the standard treatments.