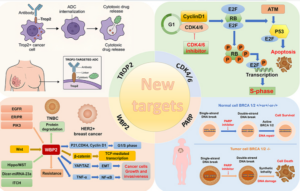
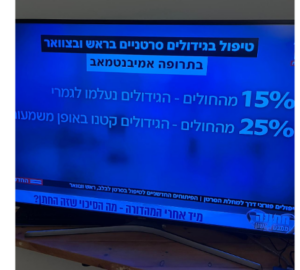
Some viruses infect and kill cancer cells.
This group of viruses is called “Oncolytic Viruses”.
They can be found in nature and can be modified in the laboratory so that they multiply in the cancer cells until they explode and then the immune system recognizes the cancer cells and attacks.
One significant benefit is that treating cancer with such a virus does not harm healthy tissues and thus saves the patient from suffering side effects related to chemotherapy.
Currently, only one Oncolytic Virus has been approved for melanoma, the genetically altered herpes virus, called talimogene laherparepvec (Imlygic®) or T-VEC. The T-VEC is injected directly into tumors and engineered to produce a protein that stimulates cells of the immune system and reduces the risk of causing herpes.
In some patients treated with T-VEC without direct injection to the tumors, the tumors shrunk so the immune system attacked the cancer even without direct injection.
There are a number of other viruses that are being studied in clinical trials to treat cancer, such as PVS-RIPO, which appears to be a major source for patients with a glioblastoma brain tumor and will soon begin a study in which GBM patients will receive this treatment with or without a chemotherapy drug.
Oncolytic Viruses are a tool that kills the cancer directly. However, more and more evidence is accumulating that they also stimulate the immune system against the cancer, and this also encourages the elimination of cancer cells. Therefore, some cancer researchers around the world call them “immunotherapy.”
Hence, there is a rationale for combining oncology with immunotherapy drugs.
There are already impressive and promising results from two such clinical trials:
one study examined nearly 200 melanoma patients who all received an immunotherapy drug called Ipilimumab® or Yervoy®, and half received T-VEC.
A second study is of 21 melanoma patients treated with a combination of an Oncolytic Virus and Keytruda®.
In light of the success, the US Food and Drug Administration (FDA) has approved a clinical trial that will soon begin, combining Keytruda® with an Oncolytic Virus with 600 melanoma patients.
To read more:
https://www.cancer.gov/…/…/oncolytic-viruses-to-treat-cancer