BACKGROUND
Cisplatin-based combination chemotherapy remains the standard of care for locally advanced or metastatic bladder cancer.
However, toxicity is substantial, responses are rarely durable, and many locally advanced or metastatic patients are ineligible.
Padcev and Keytruda have each shown a survival benefit versus chemotherapy in Bladder Cancer, are not restricted by cisplatin eligibility, and warrant investigation as a first-line combination therapy in patients ineligible for cisplatin.
WHAT IS PADCEV?
Padcev, also called Enfortumab Vedotin, is an antibody-drug conjugate used for the treatment of bladder cancer.
The most common side effects include fatigue, peripheral neuropathy, decreased appetite, rash, alopecia (hair loss), nausea, altered taste, diarrhea, dry eye, pruritus (itching) and dry skin.
The U.S. Food and Drug Administration (FDA) considers it to be a first-in-class medication.
WHAT IS KEYTRUDA?
Keytruda, also called Pembrolizumab, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer, cervical cancer, and certain types of breast cancer.
Common side effects include fatigue, musculoskeletal pain, decreased appetite, itchy skin (pruritus), diarrhea, nausea, rash, fever (pyrexia), cough, difficulty breathing (dyspnea), constipation, pain, and abdominal pain.
It is a type of antibody that blocks a protective mechanism of cancer cells and thereby, allows the immune system to destroy them.
It targets the PD-1 receptor of lymphocytes.
Keytruda was approved for medical use in the United States in 2014. In 2017, the US Food and Drug Administration (FDA) approved it for any unresectable or metastatic solid tumor with certain genetic anomalies.
It is on the World Health Organization’s List of Essential Medicines.
METHODS
In this Phase 1b/2 study, first-line cisplatin-ineligible locally advanced or metastatic patients received Padcev 1.25 mg/kg (Days 1 and 8) and Keytruda 200 mg (Day 1) intravenously in 3-week cycles.
RESULTS
Forty-five patients received Padcev plus Keytruda. The most common treatment-related side effects were peripheral sensory neuropathy, fatigue and alopecia.
Twenty-nine patients had difficult side effects. The most common were increased lipase, maculopapular rash and fatigue.
One death was associated with the treatment.
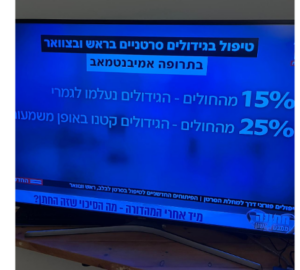
73.3% of patient benefited from the treatment experiencing a decrease in lesions’ size. In these patients the median Survival was 25.6 months.
15.6% of patients had complete remission. In these patients the median Survival was 26.1 months.
CONCLUSIONS
Padcev plus Keytruda showed a manageable safety profile.
Most patients experienced tumor shrinkage.
The median Survival exceeded 2 years in a cisplatin-ineligible patient population make this a promising combination.
Talk to us so see if we can help you to actually get the most advanced treatments
TRIAL•IN Pharma
Because we, do not give up on life!
Contact us 24/7 –
Call center +44.2082.426.039
Read more about bladder cancer >>