In mid March 2019, the international annual meeting of the Society of Gynecologic Oncology, the SGO, was held.
At the meeting, experts from the field presented their work in the areas of endometrial cancer, cervical cancer and ovarian cancer.
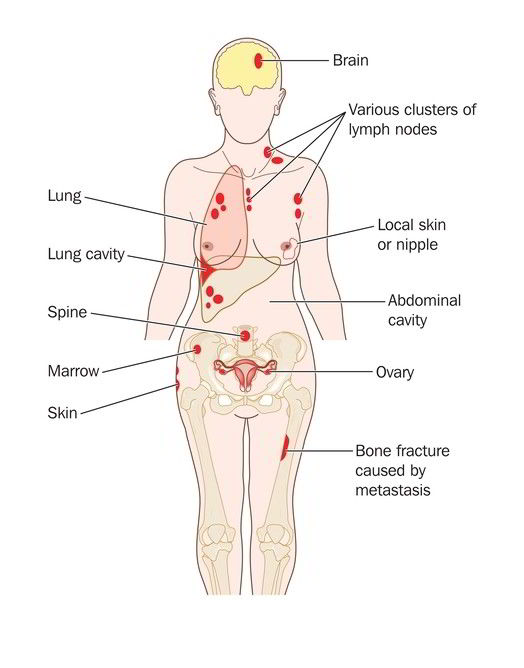
One of the most powerful studies presented, was a pilot study using combination of chemotherapy and a targeted agent in platinum-resistant ovarian cancer and recurrent endometrial cancer. The two drugs are Lenvima, which a cancer drug belongs to a family of targeted drugs and the second drug is a common chemotherapy named Taxol.
Study description
In this phase I trial, patients with recurrent endometrial and patients with platinum-resistant epithelial ovarian cancer were treated with the combination of Lenvima®, a tyrosine kinase inhibitor, and a weekly dose of Taxol.
Results
Weekly Taxol has been used for these diseases for many years with a very modest and unsatisfactory response rate.
The addition of Lenvima® to Taxol has dramatically raised treatment efficacy to overall response rate of 65% – About two third of patients responded to the combination: Their disease either stopped from growing or shrunken.
Yet, there are adverse events to the combination treatment and they are common, that may become a limitation in a larger population.
The News
The response rate was 71% in patients in the ovarian cancer group and 50% in patients with endometrial cancer.
The primary objective of the trial was to identify the right and most suitable dose of the combination for a continuing Phase II clinical trial.
In addition, secondary endpoints included follow-up on safety and tolerability, antitumor activity, pharmacokinetics and duration of treatment until further progression of disease.
To Summarize
It is almost unheard of such results in this population. Even though it was a very small trial on 26 patients, the efficacy observed was impressive!
There is no doubt that participation in Phase I clinical trials can also be of great benefit to cancer patients and by the time Phase II and Phase III clinical trials will be launched, the same patients who had joined this study – had won!