Nerlynx in metastatic cervical cancer
About a week ago, preliminary results were published in a study examining the efficacy and safety of aN approved drug, called Nerlynx. The study involved metastatic patients with cervical cancer whose cancer cells posses a mutation in a gene called HER2. The results were published at the international annual meeting of the gynecological-oncology society, the SGO.
Results
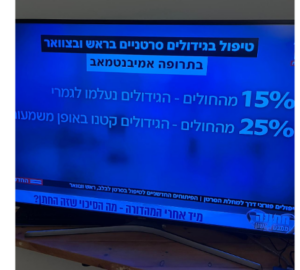
54.5% of Nerlynx patients responded: the disease was stopped or decreased in size and the mean duration of treatment with Nerlynx was 7 months.
A mutation in the HER2 gene, also known as ERBB2, is common in only 5% of this population and is generally associated with poor prognosis, which is why these results are most impressive: It is an effective treatment for severe disease. The results are even more impressive since the average number of prior treatments for their metastatic disease was two, i.e., The metastatic disease in this clinical trial is in a very advanced condition.
Adverse events
The most common adverse events among patients with Nerlynx were: diarrhea, nausea, decreased appetite, abdominal pain, shortness of breath, and others. However, no patient needed to reduce the dose or discontinue treatment entirely due to side effects.
About Nerlynx
Nerlynx is a tablet that disrupts the cancer cell division mechanism and kills it. In the clinical trial, metastatic cervical patients took a tablet of 240 mg once a day. Very convenient.
TRIAL•IN Pharma provides a unique, personalized service for patients with metastatic cancer and brain tumors, which reveals innovative and advanced cancer treatments around the world and helps to achieve them.
Call us 24/7 toll free to find out if our services match your current medical and clinical condition:
+44.2082.426.039
or leave us a message on our website and we’ll get back to you shortly:
www.trial-in.com
TRIAL•IN Pharma – We do not give up on life!