Published on February 26, 2020
Background
In heavily pretreated patients, for whom there remains a tremendous unmet need with limited options beyond single-agent chemotherapy, which generates responses in just 12% of patients with short duration and severe adverse effects.
DPX-Survivac had previously received Fast Track designation from the FDA as maintenance therapy in patients with advanced ovarian cancer.
About the DeCidE1 trial
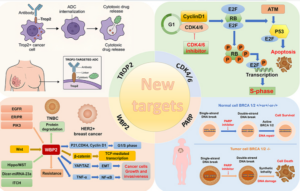
The trial which is still ongoing and recruiting, is a Phase Ib/II study evaluating the T-cell activating therapy DPX-Survivac with low-dose, oral cyclophosphamide(Cytoxan) and the IDO1 inhibitor Epacadostat in patients with recurrent ovarian, fallopian tube, or peritoneal cancer.
Updated results from the Phase Ib/II study, DeCidE1 trial, of DPX-Survivac demonstrated DPX-Survivac was active, durable and well-tolerated in patients with heavily pretreated (failed >3 lines of treatment), advanced recurrent ovarian cancer.
Patients who benefited from this agent included those with platinum-resistant and refractory disease.
Results
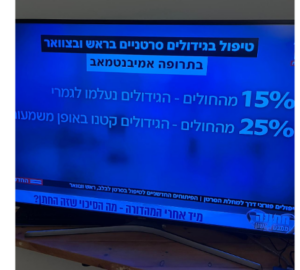
- Out of 19 patients who were evaluable for efficacy, 15 (79%) achieved disease control with either stable disease or a partial response in the target lesions.
- Ten patients (53%) demonstrated tumor shrinkage.
- Clinical benefit lasting ≥6 months was observed in 7 patients, 4 (21%) of whom achieved a partial response with >30% tumor regression on target lesions.
- 3 patients had ongoing stable disease for more than 6 months.
Side effects
Treatment was well tolerated, with most toxicity events being low in severity at the injection site and maintained quality of life.
The best treatment for a cancer patient is to get the most advanced cancer drugs in advanced stages of development. There, the hope and the chance to extend life go far beyond the standard protocols.
Contact us to find out what is the best treatment for YOU
TRIAL•IN Pharma
Because we, do not give up on life!
Contact us 24/7 for free service matching:
Call center +44.2082.426.039
About Ovarian Cancer
1 in 80 women will develop ovarian cancer during their lifetime. This is the fifth-ranked cancer-related death among women.
The ovaries are part of the female reproductive system, where the egg cells are produced for the reproductive process. The ovaries are also the main source of female hormones: Estrogen and Progesterone.
The existing therapies used for stage 4 metastatic ovarian cancer are not curative, and this is a source of the need for innovative and effective treatment strategies in order to fight the disease.
Stage 4 Metastatic Ovarian Cancer
Stage 4 metastatic ovarian cancer is a cancer that started in the ovarian and its cells have spread to other body organs, distant from the ovarian, and created new lesions. These lesions are called “metastases” and are made of ovarian cancer cells.
Ovarian cancer metastases may appear in the bones, liver, lungs and brain. The formation of metastases constitutes a threat on life.
Even though the cancer has spread to other organs, it is still defined as a ovarian cancer and is treated as ovarian cancer. In this manner for example, a lesion in the lung that is a metastasis of ovarian cancer shall be treated with ovarian cancer drugs and not lung cancer drugs.
Treatment Goals
The goal of treating local ovarian cancer, not metastatic, is to remove and eradicate the tumor completely.
In metastatic stage 4 disease, the primary objective of the treatment is to slow down and control disease progression and help reduce symptoms as well prolong life and improve patient’s quality of life.