When we were at the beginning with ‘TRIAL-IN Pharma’, I.C.’s son approached us and asked whether we can help his dad, a 58 years old US resident who was diagnosed with GBM, Glioblastoma and an aggressive form of brain tumor which originates in the brain.
One of the clinical trials we located for his individual unique condition is the one discussed in this post, but his oncologist told him: “You are not a stage of clinical trials yet”. I.C.’s son knew exactly how much time his father has with the standard protocols offered to everyone and was very much reluctant to accept what the oncologist had said, and his father – cooperated.
To make the story short, I.C. joined the clinical trial … he shattered the bleak statistics his oncologist had given to him:
Keytruda before and after surgery extends lives of GBM, Glioblastoma patients with recurrent disease
Around mid February 2019, results were published from a randomized clinical trial which was held in 7 cancer centers in the US and looked at the immune responses and overall survival of a treatment with Keytruda, before and/or after surgical procedure among 35 recurrent GBM patients whose recurrent GBM is resectable (operable).
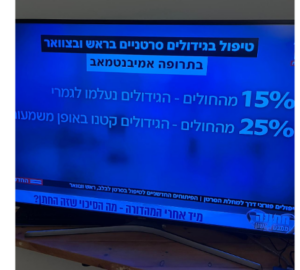
The group of patients that received Keytruda before AND after surgery lived almost twice as much (!) compared to the group of patients who was operated and then received Keytruda. Additional findings were that the risk for death during the treatment dropped by 61% among the group of patients that received Keytruda before AND after surgery!
Further observations were detected in which the immune system activity against the cancer was impressively raised both systemically and locally in the tumor microenvironment.
Keytruda is FDA approved for various indications.
TRIAL-IN Pharma offers a unique, personalized research and analysis for patients with metastatic cancer and brain tumors, which reveals innovative and advanced oncologic treatments around the world and supports the process of achieving them.
For free service evaluation, dial 24/7
+44.2082.426.039
Do not give up on life!
For further reading
https://www.ncbi.nlm.nih.gov/pubmed/30742122
https://www.clinicaltrials.gov/ct2/show/NCT02852655?term=pembrolizumab+recurrent+glioblastoma&rank=8