Importance
Endometrial cancer is often hormone-dependent and treated with aromatase inhibitors, (AIs).
The uncontrolled PI3K-AKT-mTOR pathway observed in this type of cancer drives hormonal resistance. Therefore, there is rationale of combining mTOR inhibitor with endocrine therapy.
This is what the investigators evaluated in the VICTORIA clinical trial.
This phase I/II clinical trial evaluated the efficacy and safety of Vistusertib, an mTOR inhibitor, combined with Arimidex in patients with hormone receptor–positive, HR+, recurrent or metastatic endometrial cancer.
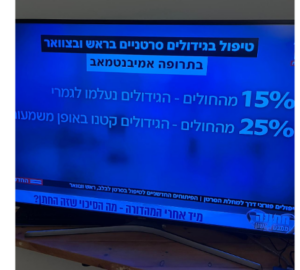
After 8 weeks the percentage of women who had no progression almost doubled with Vistusertib plus Arimidex compared with Arimidex alone.
Vistusertib plus Arimidex demonstrated clinically meaningful benefit, with manageable side effects, in patients with recurrent or metastatic HR+ endometrial cancer.
Objective
To evaluate the safety and efficacy of Vistusertib in combination with Arimidex in the treatment of women with hormone receptor-positive (HR+) recurrent or metastatic endometrial cancer.
About the VICTORIA Study in Endometrial Cancer Patients
The VICTORIA study was a multicenter, randomized clinical trial that enrolled 75 patients with hormone receptor-positive recurrent or metastatic endometrial cancer.
Patients were recruited to the study from 12 cancer centers in France in April 2016 to October 2019.
Study Treatment
Patients were randomized to two arms: Oral Vistusertib (125 mg twice daily 2 days per week) and oral Arimidex (1 mg daily) in the combination Vistusertib with Arimidex arm (V+A arm) or oral Arimidex alone (A arm).
Results of the Study
Of the 75 patients, 73 were treated:
V+A arm: 49 patients
A arm: 24 patients
In the V+A arm, 67.3% of patients were did not progress after 8 weeks and in the A arm, 39.1% did not progress.
No significant serious side effects were reported.
The overall Response Rate was 24.5% in the V+A arm vs 17.4% in the A arm.
Median Progression Free Survival, PFS, was 5.2 months in the V+A arm and 1.9 months in the A arm.
Fatigue, lymphopenia, hyperglycemia, and diarrhea were the most common side effects associated with Vistusertib.
Conclusions for Endometrial Cancer Patients
This multicenter, phase 1/2 clinical trial demonstrated that adding Vistusertib to Arimidex improved treatment outcomes for patients with endometrial cancer.
Vistusertib plus Arimidex had manageable side effects.
Talk to us so see if we can help you to actually get the most advanced treatments
TRIAL•IN Pharma
Because we, do not give up on life!
Contact us 24/7 –
Call center +44.2082.426.039